Doctor Doping Shop
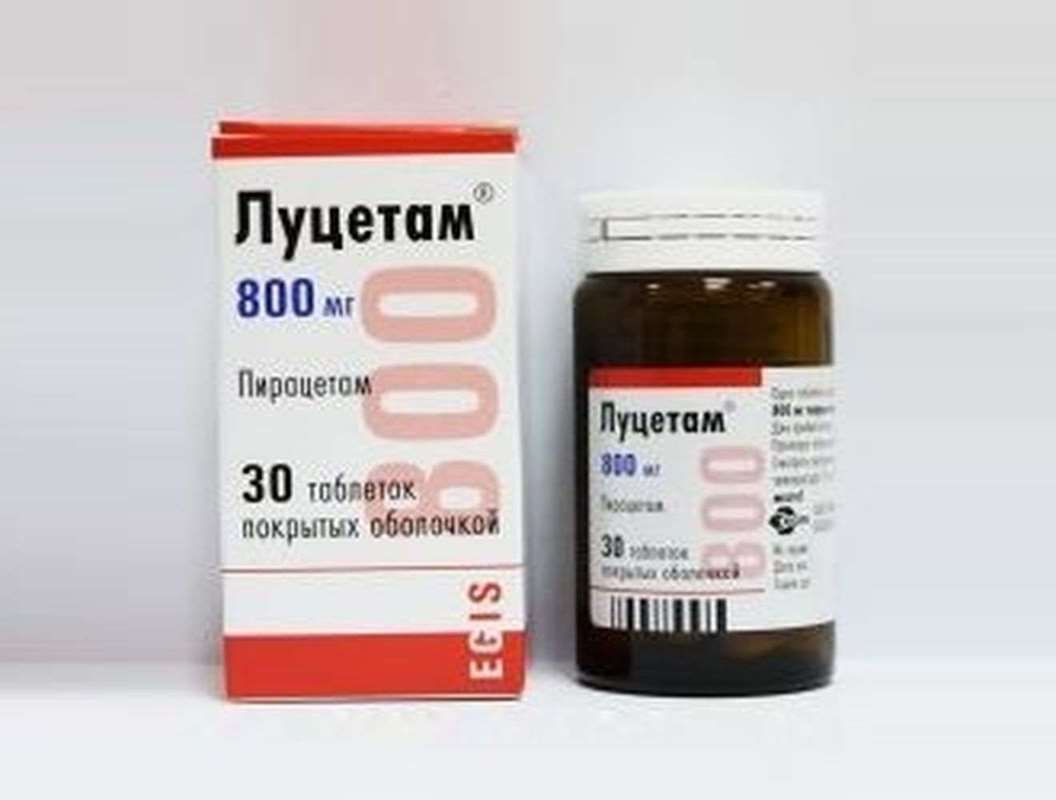
Lucetam 800mg 30 pills
USD 12.00
In stock
Be the first to review this item
Lucetam (Piracetamum, Piracetam) - neuroprotective drugs.
Piracetam - cyclic derivative of GABA. Direct impact on the brain. Lucetam improves cognitive (cognitive) processes, such as learning ability, memory, attention, ability to memorize, as well as improves mental performance, without the development of sedative and psychostimulant effect.
Has impact on the central nervous system in various ways: change the speed of propagation of excitation in the brain, improves neuronal plasticity and metabolic processes in the nerve cells. It improves the interaction between the hemispheres of the brain and synaptic conductance in neocortical structures, it improves mental performance, improves cerebral blood flow.
Lucetam improves microcirculation in the brain, affecting the rheological properties of blood and causes vasodilatory action. Lucetam inhibits platelet aggregation and restores the elasticity of erythrocyte membranes, as well as the ability of the latter to the passage through the microcirculation. It reduces adhesion of red blood cells. At a dose of 9.6 g reduces levels of fibrinogen and von Willebrand factor by 30-40% and prolongs bleeding time.
Lucetam has a protective and restorative action in violation of brain function due to hypoxia, intoxication or injury. It reduces the severity and duration of vestibular nystagmus.
Testimony:
- Symptomatic treatment of psycho-organic syndrome, particularly in elderly patients suffering from memory loss, concentration and total activity, dizziness, mood changes, conduct disorder, gait disorder, as well as in patients with Alzheimer's disease and senile dementia of the Alzheimer type.
- Treatment of the consequences of ischemic stroke as speech disorders, disorders of the emotional sphere, motor and mental activity.
- Chronic alcoholism - for the treatment of psycho-organic and withdrawal syndromes.
- During the recovery period after injuries and cerebral intoxication.
- Treatment of vertigo and associated disorders of balance, with the exception of dizziness psychic origin.
- In the complex treatment of lower learning in children, especially in the case of difficulty of acquisition of specific skills of reading, writing, numeracy, which can not be explained by mental retardation, inadequate training or feature family environment.
- For the treatment of cortical myoclonus as mono- or combination therapy.
Contraindications:
- Hemorrhagic stroke.
- End-stage renal failure (creatinine clearance at least 20 ml / min).
- Children under the age of 1 year.
- Pregnancy.
- Lactation.
- Hypersensitivity to piracetam or pyrrolidone derivatives, and other ingredients.
Pregnancy and breast-feeding:
Animal studies have not revealed the damaging effect on the embryo and the development of the offspring, including in the postnatal period, and did not change during pregnancy and childbirth. Studies have not been conducted in pregnant women. Piracetam crosses the placental barrier and into breast milk. The concentration of the drug in infants up to 70-90% of its concentration in the blood of the mother. Except in special circumstances Lutsetam should not be administered during pregnancy. It should refrain from breast-feeding in the appointment of a woman Lutsetam.
Side effects:
Disorders of the nervous system: hyperkinesis, irritability, drowsiness, depression, fatigue: these symptoms are more common in elderly patients receiving a dose of more than 2.4 g / day, in most cases it is possible to achieve regression of these symptoms by reducing the dose. Headache, dizziness, insomnia, mental excitement, anxiety, balance disorder, tremor, ataxia, exacerbation of epilepsy, anxiety, hallucinations, confusion, increased libido.
- Violations by the blood and lymph: bleeding
- Violations of the immune system: hypersensitivity, including anaphylaxis
- Violations of the organ of hearing and the labyrinth: dizziness (vertigo)
- Violations of the cardiovascular system: decrease or increase arterialnogodavleniya.
- Disorders of the digestive system: nausea, vomiting, diarrhea, abdominal pain, epigastric pain.
- Violations of the Metabolism and nutrition: increasing body mass.
- Violations of the skin: dermatitis, pruritus, urticaria.
- Allergic reactions: angioedema.
- General disorders and injection site: pain at the injection site, thrombophlebitis, pyrexia.
Overdose:
Treatment: Immediately after ingestion of the drug, you can wash out the stomach or cause an artificial vomiting. Conduct symptomatic therapy, which may include hemodialysis. No specific antidote. The effectiveness of hemodialysis for piracetam is 50-60%.
Special instructions:
In connection with the effect of piracetam on platelet aggregation caution should be prescribed to patients with impaired hemostasis during major surgery or in patients with symptoms of severe bleeding.
In the treatment of cortical myoclonus abrupt interruption of treatment should be avoided, which might cause the resumption of seizures.
It penetrates through the filtration membranes for hemodialysis machines.
When long-term therapy of elderly patients are advised to regularly monitor indicators of kidney function, if necessary, shall adjust the dose depending on the results of research spacecraft.
Taking into account possible side effects, the patient should be careful when driving and operating machinery.
Suggested Use:
Inside the drug is administered in a daily dose of 30-160 mg / kg, the multiplicity of reception - 2-4 times / day.
Lutsetam tablets taken with meals or on an empty stomach, with some liquid (water, juice).
administered 1.2-2.4 g / day, and during the first week in the symptomatic treatment of chronic psycho-organic syndrome depending on the severity of symptoms - 4.8 g / day.
In the treatment of the effects of stroke appoint 4.8 g / day.
When alcohol withdrawal syndrome - 12 g / day. The maintenance dose - 2.4 g / day.
For the treatment of vertigo and associated disorders of balance administered 2.4-4.8 g / day.
When cortical myoclonus treatment starts with a dose of 7.2 g / day, once every 3-4 days, increase the dose of 4.8 g / day to a maximum dose of 24 g / day. Treatment continued throughout the period of the disease. Every 6 months should attempt to reduce the dose or stop the drug, gradually reducing the dose of 1.2 g every 2 days in order to prevent an attack. In the absence of treatment effect or a slight therapeutic effect is stopped.
Babies for the correction of learning disabilities prescribed oral dose of 3.2 g / day. Treatment continues throughout the school year.
In patients with impaired function of the liver correction mode is not required.
In patients with impaired renal function requires correction dosing regimen according to creatinine clearance (CC). The degree of renal failure QC (ml / min) Dose Rate of> 80 Usual dose Light 50-79 2/3 usual dose is 2 - 3 admission average 30-49 1/3 usual dose in 2 hours Weight <30 1/6 usual dose, End-stage single - contraindicated
In elderly patients a dose of correcting the presence of renal insufficiency and long-term treatment is necessary to monitor renal function.
Drug interactions:
While the use of thyroid extract can be irritability, disorientation and sleep disorders.
There was no interaction with clonazepam, phenytoin, phenobarbital, sodium valproate.
Piracetam in high doses (9.6 g / d) increases the efficiency of acenocoumarol in patients with venous thrombosis (indicated a greater reduction in the level of platelet aggregation, fibrinogen, von Willebrand factor, blood and plasma viscosity in comparison with the use of only acenocoumarol).
Ability to change the pharmacodynamics piracetam under the influence of other drugs is low because 90% of the drug is excreted unchanged in the urine.
In vitro piracetam does not inhibit isozymes CYP1A2, 2B6, 2S8, 2C9, 2C19, 2D6, 2E1, and 4A9 / 11 in a concentration of 142, 426 and 1422 pg / mL. At a concentration of 1422 mg piracetam / ml there was a slight inhibition of CYP2A6 (21%) and 3A4 / 5 (11%). However, the level of the two isoenzymes Ki sufficient in excess of 1422 mg / ml. Therefore, metabolic interactions with other drugs are unlikely.
Receiving Piracetam 20 mg / day did not change the level curve and peak concentration of antiepileptic drugs in serum (carbamazepine, phenytoin, phenobarbital, valproate) in epileptic patients receiving the drug in a constant dose.
Co-administration with ethanol did not affect the level of concentration of piracetam serum concentration of ethanol in serum did not change when receiving 1.6 g piracetam.
Packaging:
- Comes in original packaging. Item is brand new and unopened.
Storage:
- Keep away from direct sunlight.
- Keep locked and away from children.
- Store in dry place at room temperature.
- Do not exceed storage temperature higher than 25 C
Important notice- the outer box design may vary before prior notice!
Related products


 Cart
Cart