Doctor Doping Shop
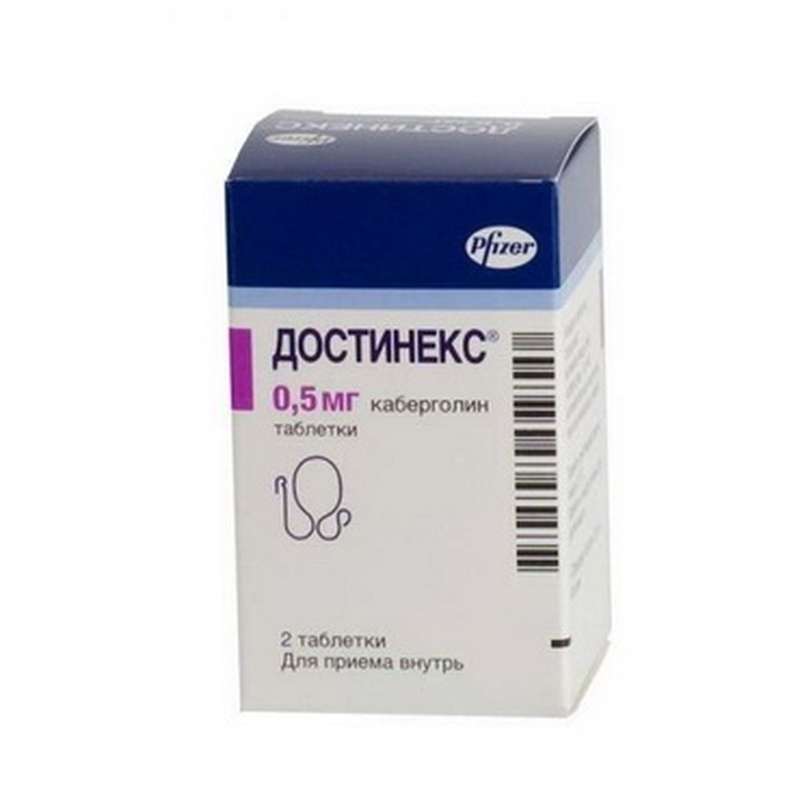
Dostinex 0.5mg 2 pills
USD 55.00
In stock
Be the first to review this item
Dostinex is hypoprolactinemic.
Cabergoline is a dopaminergic derivative of ergoline and is characterized by a pronounced and prolonged prolactin-lowering effect due to direct stimulation of the D2-dopamine receptors of lactotrophic pituitary cells. In addition, when taking higher doses compared to doses to reduce serum prolactin levels, cabergoline exhibits a central dopaminergic effect due to stimulation of D2 receptors.
Reduction in the concentration of prolactin in the blood plasma is observed within 3 hours after taking the drug and persists for 7-28 days in healthy volunteers and patients with hyperprolactinemia and up to 14-21 days - in women in the postpartum period.
Cabergoline has a strictly selective effect, does not affect the basal secretion of other hormones of the pituitary and cortisol. Prolactin-lowering effect of the drug is dose-dependent, both in terms of severity and duration of action.
The pharmacodynamic effects of cabergoline, not related to the therapeutic effect, include only a reduction in blood pressure. With a single admission of the drug, the maximum antihypertensive effect occurs within the first 6 hours and is dose-dependent.
Indications:
- prevention of physiological postpartum lactation (only for medical reasons);
- suppression of already established lactation (only for medical reasons);
- treatment of disorders associated with hyperprolactinaemia, including functional disorders such as amenorrhea, oligomenorrhoea, anovulation and galactorrhea;
- Prolactin-secreting adenomas of the pituitary gland (micro- and macro-prolactinomas, idiopathic hyperprolactinemia or the syndrome of the "empty" Turkish saddle in combination with hyperprolactinemia.
Contraindications:
- postpartum arterial hypertension;
- preeclampsia;
- pregnancy;
- increased sensitivity to cabergoline or other components of the drug, as well as to any ergot alkaloids.
Suggested Use:
Dostinex should be taken orally, preferably during meals.
To prevent lactation appoint the drug in a dose of 1 mg (2 tablets) once on the first day after childbirth.
To suppress the established lactation, 0.25 mg (1/2 tablet) is prescribed 2 times a day for 2 days (the total dose is 1 mg).
To treat disorders associated with hyperprolactinemia, the drug is prescribed at a dose of 0.5 mg per week in 1 or 2 doses (1/2 tablet, for example, on Monday and Thursday). Increase the weekly dose should be carried out gradually - by 0.5 mg at intervals of 1 month to achieve the optimal therapeutic effect. The average therapeutic dose is 1 mg per week, but can range from 0.25 mg to 2 mg per week. Patients with hyperprolactinemia use doses up to 4.5 mg per week.
When appointing the drug at a dose of 1 mg per week or more, the drug should be divided into 2 or more receptions per week, depending on the tolerability.
Special instructions:
After selecting an effective dosing regimen, it is recommended to conduct a regular (1 time per month) determination of serum prolactin level. Normalization of prolactin levels is usually observed within 2-4 weeks of treatment.
Dostinex with caution is prescribed for patients with cardiovascular diseases, Raynaud's syndrome, with peptic ulcer or gastrointestinal bleeding, as well as patients with indications of a history of severe mental illness, especially psychoses.
With caution appoint Dostinex on the background of therapy with drugs that have hypotensive effect.
With prolonged therapy, Dostinex should be administered at lower doses to patients with severe hepatic insufficiency.
After the abolition of Dostinex, a relapse of hyperprolactinemia is usually observed. However, in a number of patients, persistent suppressive levels of prolactin have been observed for several months. Most women register ovulatory cycles for at least 6 months after the abolition of Dostinex.
Do not exceed a single dose of Dostinex equal to 0.25 mg in nursing mothers undergoing treatment aimed at suppressing the already established lactation in order to prevent the occurrence of orthostatic hypotension.
Before the start of therapy, Dostinex showed a complete examination of the pituitary function.
Dostineks restores ovulation and fertility in women with hyperprolactinemic hypogonadism. Because pregnancy can occur before the recovery of menstruation, it is recommended that pregnancy tests be performed at least once every 4 weeks during the amenorrhea period, and after the recovery of menstruation, whenever there is a delay in menstruation for more than 3 days. For women who want to avoid pregnancy, for the period of treatment with Dostinex, and after the abolition of Dostinex and before the return of anovulation, nonhormonal methods of contraception should be used.
Women who have become pregnant should be under the supervision of a doctor to detect symptoms of pituitary gland enlargement in time, since during pregnancy it is possible to increase the size of pre-existing pituitary tumors.
Adverse events associated with taking the drug, are dose-dependent. The likelihood of developing side effects can be reduced by initiating therapy with Dostinex in lower doses (for example, at 0.25 mg once a week), followed by a gradual increase in the dose until the therapeutic dose level is reached. If there are persistent or severe side effects, then a temporary dose reduction, and then a more gradual increase (for example, an increase of 0.25 mg per week every 2 weeks) will contribute to better tolerability of the drug.
With prolonged therapy, Dostinex does not exhibit a deviation from the norm of standard laboratory tests; in women with amenorrhea, there was a decrease in hemoglobin levels during the first few months after the recovery of menstruation.
Side effects:
From the CCC: palpitation; rarely - orthostatic hypotension (with prolonged use of Dostinex � usually has an antihypertensive effect); possibly asymptomatic decrease in blood pressure during the first 3-4 days after birth (SAD - more than 20 mm Hg, DAD - more than 10 mm Hg).
From the nervous system: dizziness / vertigo, headache, fatigue, drowsiness, depression, asthenia, paresthesia, fainting.
On the part of the digestive system: nausea, vomiting, pain in the epigastric region, abdominal pain, constipation, gastritis, dyspepsia.
Other: mastodinia, epistaxis, flushing of the blood to the skin of the face, transient hemianopia, spasms of the vessels of the fingers and cramps of the muscles of the lower extremities (like other ergot derivatives, Dostinex� may have a vasoconstrictive effect).
Packaging:
- Comes in original packaging. Item is brand new and unopened.
Storage:
- Keep away from direct sunlight.
- Keep locked and away from children.
- Store in dry place at room temperature.
- Do not exceed storage temperature higher than 25 C
Important notice- the outer box design may vary before prior notice!
Related products


 Cart
Cart