Doctor Doping Shop
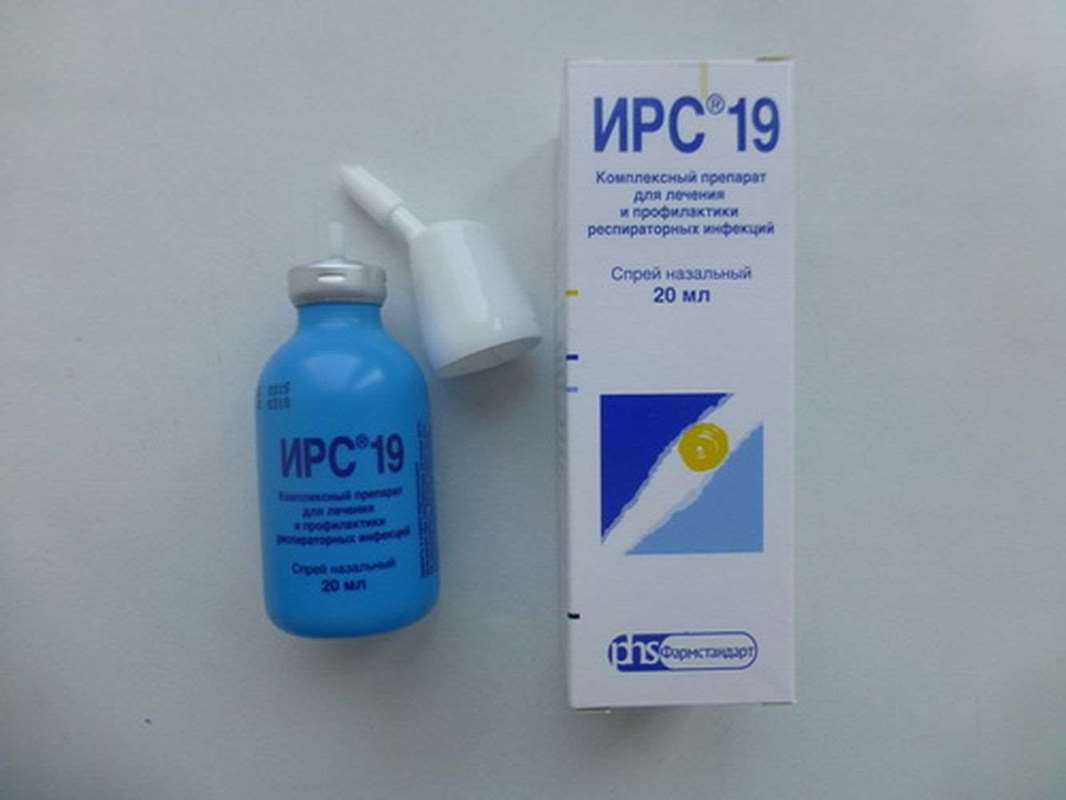
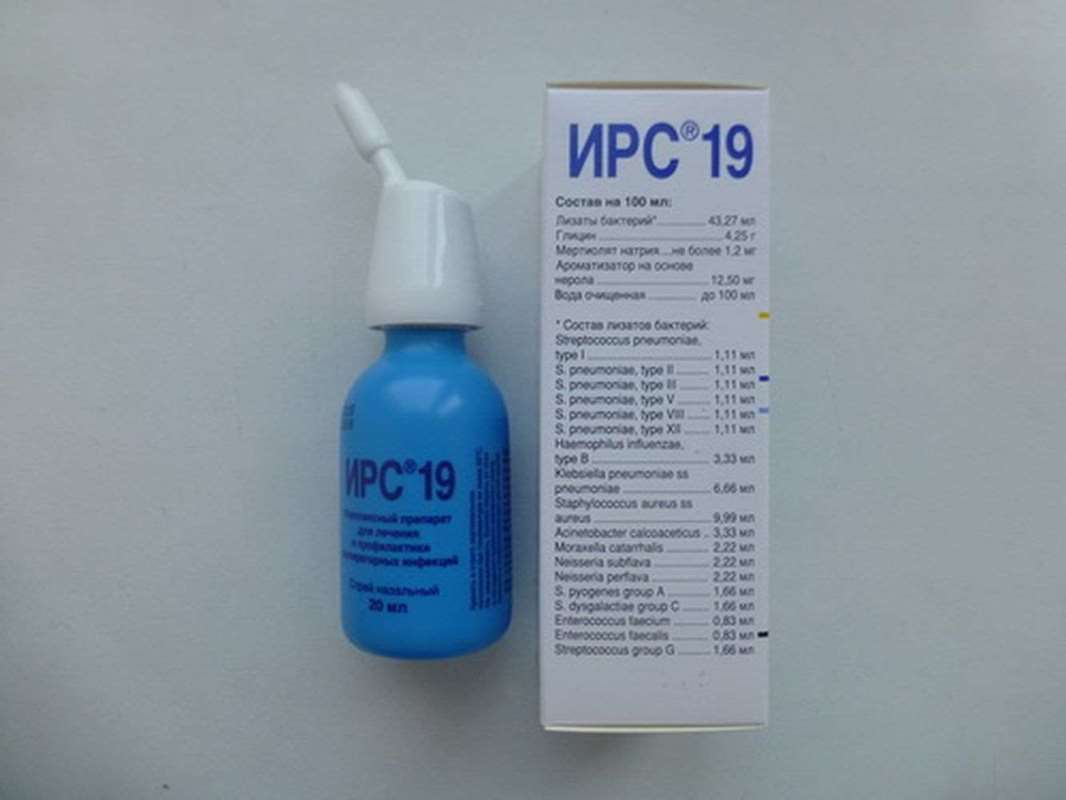
IRS 19 complex nasal spray 20ml
USD 45.00
In stock
Be the first to review this item
Complex nasal spray helps recover IRS 19 2 times faster and reduce the risk of acute respiratory infections during epidemics. It is well known that respiratory infections are spread mainly by droplets. This means that to protect themselves from them is extremely difficult - just go to the store or take public transport, and you are already at risk. Effective decision - to restore local immunity of the nasal mucosa using IRS19 spray for children and adults.
By activating antiviral and antibacterial mechanisms protection, IRS 19 c helps fight the cause of the disease. IRS 19 well tolerated, approved for use in children from 3 months and adults. 1 bottle is enough for one course of treatment or profilaktiki.
IRS 19 heals and protects from respiratory infections
IRS 19 - Immunomodulatory Respiratory Spray - heals and protects against respiratory infections. When spraying IRS 19 provides an effective immune response in the mucosa of the nose, there where infection falls first of all - in the airways. IRS19 helps to remove pathogens with the mucosal surface, maintaining the normal flora of the respiratory tract and strengthening immuniteta.
Indications
Adults and children over 3 months:
- Prevention of chronic diseases of the upper respiratory tract and bronchi;
- Treatment of acute and chronic diseases of the upper respiratory tract and bronchi, such as rhinitis, sinusitis, laryngitis, pharyngitis, tonsillitis, tracheitis, bronchitis, and other;
- Restoring local immunity post-influenza or other viral infections;
- Preparation for the planned surgery on the upper respiratory tract and the postoperative period.
When spraying IRS 19 covers the mucous membrane in the nose and provides an effective immune response at the site where the infection penetrates immediately, ie breathing putyah.
IRS 19 helps to remove pathogens with the mucosal surface, maintaining the normal flora of the respiratory tract and strengthening immuniteta.
Active substance: bacterial lysates - 43.27 ml: Streptococcus pneumoniae type I, II, III, V, VIII, XII - no 1,11 ml; Staphylococcus aureus ss aureus - 9,99 ml; Neisseria subflava - 2,22 ml; Neisseriaperflava -2,22 ml; Klebsiella pneumoniae ss pneumoniae - 6,66 ml; Moraxella catarrhalis - 2,22 ml; Haemophilus influenzae type B - 3.33 ml; Acinetobacter calcoaceticus - 3,33 ml; Enterococcus faecium - 0,83 ml; Enterococcus faecalis - 0,83 ml; Streptococcus pyogenes group A - 1,66 ml; Streptococcus dysgalactiae group C - 1,66 ml; Streptococcus group G - 1,66 ml.
Excipients: glycine, sodium thimerosal, flavor, purified water to 100 ml; vial - 20 ml (60 doses). Clear, colorless, sometimes with a yellowish tint liquid with a faint odor of flavor on the basis of nerol.
Side effect:
During the reception may experience side effects such as related and unrelated to the action of the drug.
Skin reactions: In rare cases, hypersensitivity reactions (urticaria, angioedema) and skin and eritemopodobnye ekzemopodobnye reaction.
On the side of upper respiratory tract and the respiratory system: rarely asthma and cough.
In rare cases, early treatment may experience fever (> 39 � C) for no apparent reason, nausea, vomiting, abdominal pain, diarrhea, nasopharyngitis, sinusitis, laryngitis, bronchitis.
Described isolated cases of the appearance of thrombocytopenic purpura, and erythema nodosum.
When the above symptoms it is recommended to consult a doctor.
Suggested Use:
The drug is used intranasally by aerosol administration of one dose (1 dose = 1 short pressure sprayer).
In order to prevent adults and children from 3 months to 1 administered a dose of the drug in each nostril 2 times / day for 2 weeks (to start a course of treatment is recommended for 2-3 weeks before the expected rise of morbidity).
For the treatment of acute and chronic diseases of the upper respiratory tract and bronchi to children aged from 3 months to 3 years appoint 1 dose of the drug in each nostril 2 times / day after the preliminary release of mucous discharge to the disappearance of symptoms of infection; children over 3 years of age and adults - 1 dose of the drug in each nostril 2 to 5 times / day until the disappearance of symptoms of the infection.
To restore local immunity post-influenza and other respiratory viral infections for children and adults appoint 1 dose of the drug in each nostril 2 times / day for 2 weeks.
In preparation for the planned surgery and in the postoperative period in adults and children administered 1 dose of the drug in each nostril 2 times / day for 2 weeks (to start a course of treatment is recommended for 1 week before the planned surgery).
Packaging:
- Comes in original packaging. Item is brand new and unopened.
Storage:
- Keep away from direct sunlight.
- Keep locked and away from children.
- Store in dry place at room temperature.
- Do not exceed storage temperature higher than 25 C
Important notice- the outer box design may vary before prior notice!


Related products


 Cart
Cart