Doctor Doping Shop
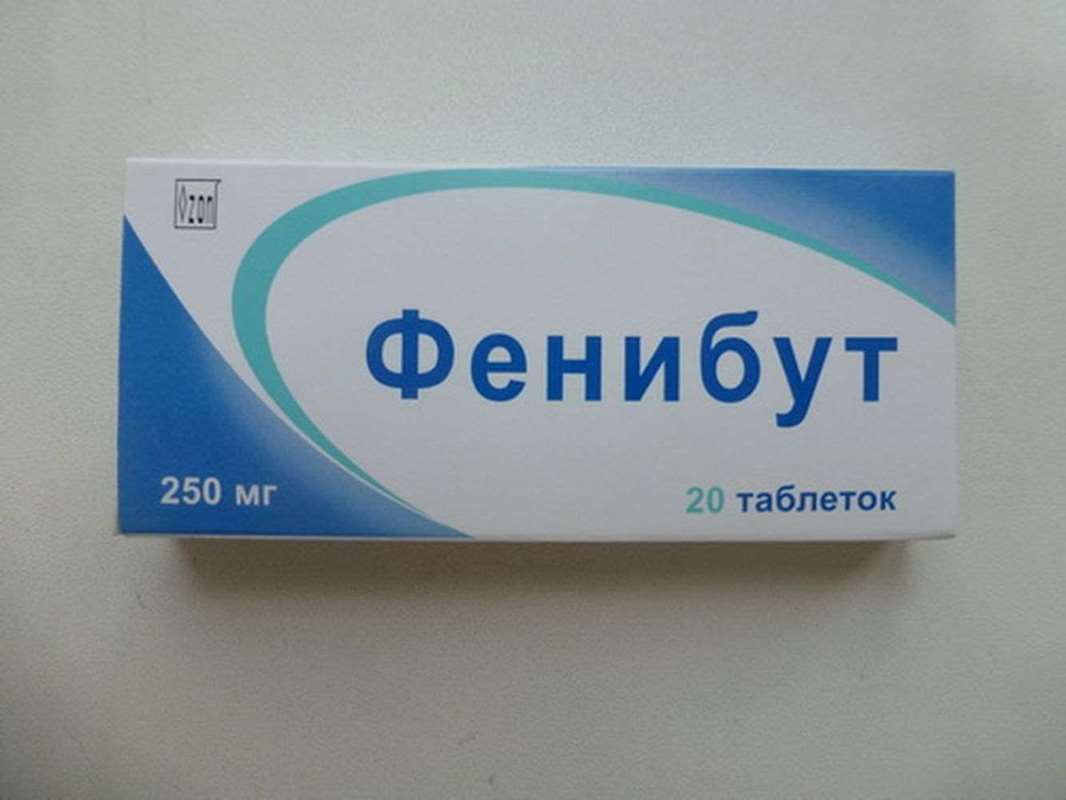
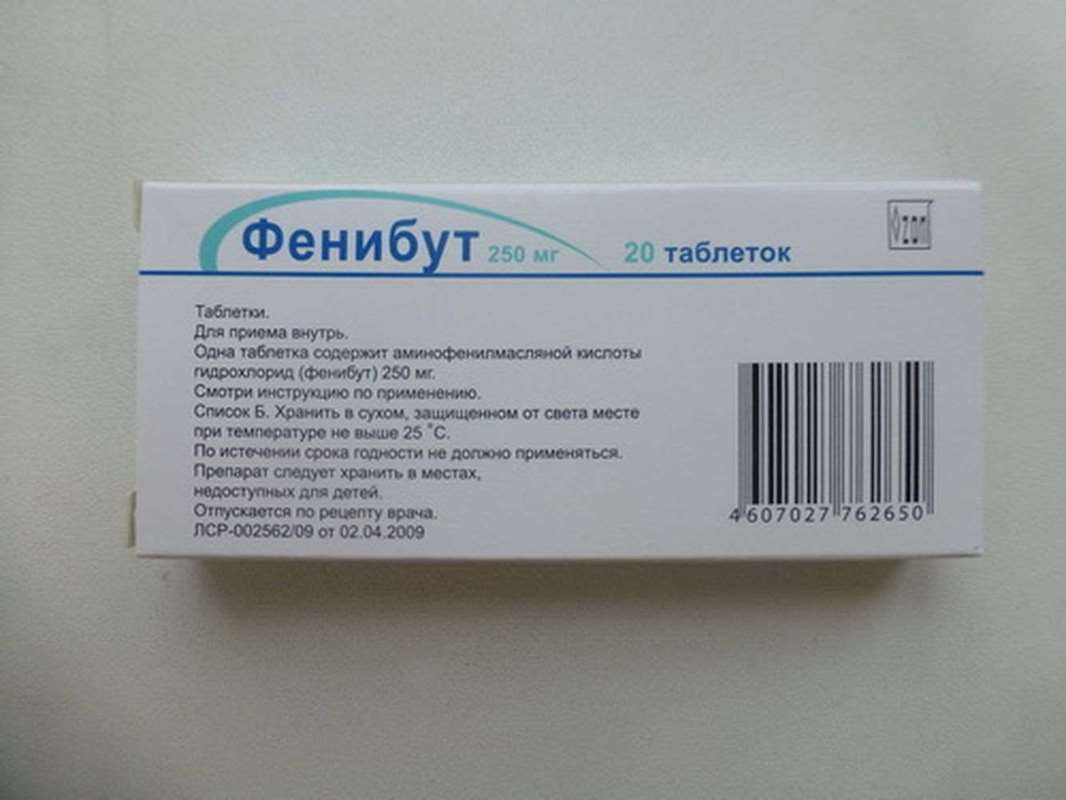
Phenibut 250mg 20 pills
USD 70.00
Out of stock
Phenibut (Phenybut) a positive effect on metabolic processes in the nerve cells of the brain. Phenibut active substance (γ-β-amino-phenylbutyric acid hydrochloride) can be regarded as a derivative of γ-amino butyric acid (GABA) derivative or β-phenylethylamine. Phenibut possesses nootropic activity, and as a derivative of GABA has anxiolytic (tranquilizing) action. No effect on choline and adrenergic receptors.
The drug reduces tension, anxiety, fear, and improves sleep, so it is used for the treatment of neuroses and before operations. Phenibut lengthens and strengthens the effects of sleeping pills, narcotic, neuroleptic and antiparkinsonian agents. Phenibut devoid of anticonvulsant activity. Phenibut extend the latency period of nystagmus and shortens its duration and severity. The drug significantly reduces the symptoms of fatigue and vasovegetative symptoms, including headache, a feeling of heaviness in the head, sleep disturbances, irritability, emotional lability, and improves mental performance. Psychological performance (attention, memory, speed and accuracy of sensory-motor reactions) under the influence of phenibut improved, unlike the influence of tranquilizers. In patients with asthenia and emotionally labile patients from the first days of therapy improved subjective well-being, increased interest and initiative, motivation activities without undue sedation or excitation.
Pharmacokinetics
Absorption - high penetrates well in all body tissues and the blood brain barrier (about 0.1% to penetrate the brain tissue of the administered dose of the drug, and in young and elderly patients are much more). Evenly distributed in the liver and kidneys. It is metabolized in the liver - 80-95%, the pharmacologically inactive metabolites. Not accumulates. After 3 hours, the kidney begins to stand out. Approximately 5% is excreted by the kidneys unchanged, partly - with bile.
Indications:
- asthenic and anxious-neurotic condition;
- worry, anxiety, fear;
- insomnia;
- as sedative before surgical interventions;
- Meniere's disease;
- dizziness associated with dysfunction of the vestibular apparatus;
- Prevention of motion sickness;
- stuttering in children and teak.
- There is evidence of increased under the influence of anti-Parkinsonian action phenibut funds (due to the presence of dofaminopozitivnogo component of the drug).
Side effect:
Phenibut usually well tolerated. At the first receptions phenibut or overdose can occur drowsiness.
Suggested Use:
Inside, after eating, adults - 0.25-0.5 g 3 times a day courses for 2-3 weeks. If necessary, increase the dose to 0.75 g (persons over 60 years appoint not more than 0.5 g per reception).
Children up to 8 years - 0.05-0.1 g per reception; from 8 to 14 years - 0.25 g per reception. For treatment of alcohol withdrawal syndrome are administered in the first few days of treatment at 0.25-0.5 g 3 times during the day and 0.75 g for the night, gradually reducing the dose. For prevention of motion sickness is prescribed for 1 hour before the expected start of the attack, or at the first sign, once at 0.25-0.5 g After the onset of motion sickness pronounced effects (vomiting, etc..), The drug is ineffective.
Packaging:
- Comes in original packaging. Item is brand new and unopened.
Storage:
- Keep away from direct sunlight.
- Keep locked and away from children.
- Store in dry place at room temperature.
- Do not exceed storage temperature higher than 25 C
Important notice - the outer box design may vary before prior notice!
Related products


 Cart
Cart