Doctor Doping Shop
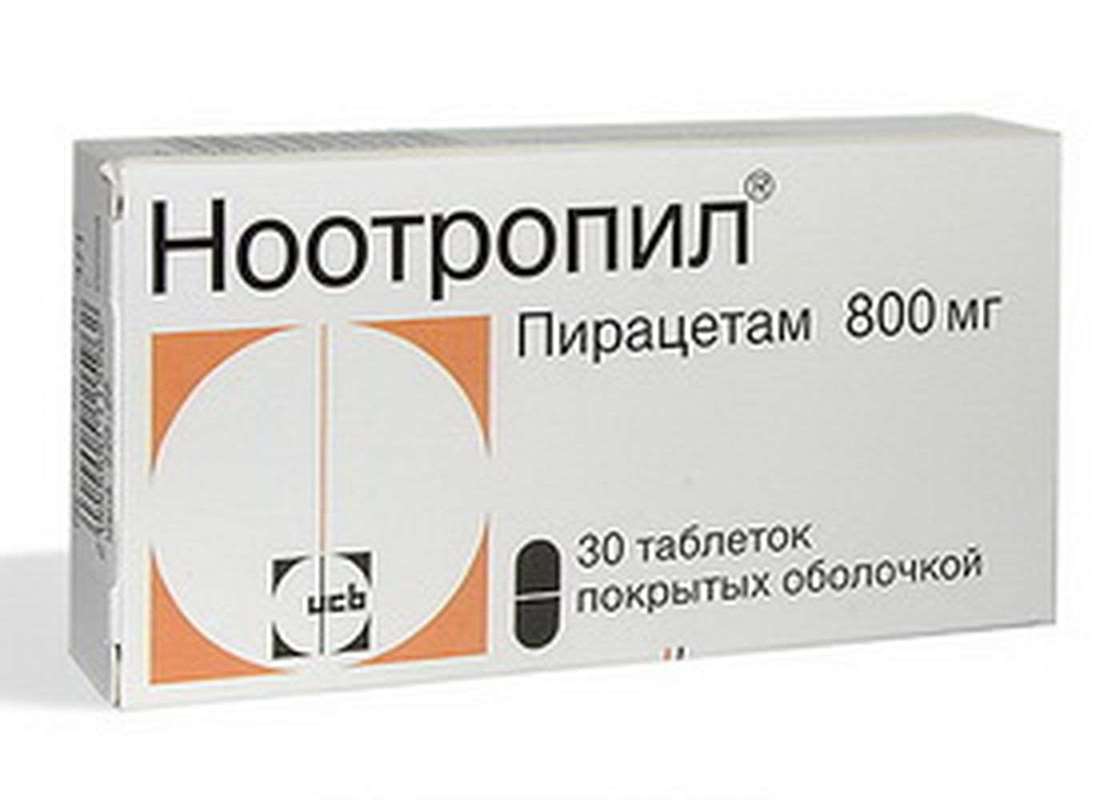
Nootropil 800mg 30 pills
USD 24.00
In stock
Be the first to review this item
Nootropil - nootropic agent.
Active ingredient - Piracetam, a cyclic derivative of GABA.
Piracetam - nootropic drug that directly improves brain function performed. The drug has an effect on the central nervous system in different ways: modifies neurotransmission in the brain, improves the metabolic conditions that promote neuronal plasticity, improves microcirculation by acting on the rheological properties of blood and causing vasodilation.
Long or short the use of piracetam in patients with cerebral dysfunction increases concentration and improves cognitive function, as manifested by significant changes in the EEG (increased α and β activity, decreased activity of δ).
The drug helps restore cognitive abilities after various cerebral damage due to hypoxia, intoxication, or electroconvulsive therapy. Piracetam is indicated for the treatment of cortical myoclonus, both as monotherapy and as part of combination therapy.
It reduces the duration of vestibular neyronita provoked.
Piracetam increased inhibits aggregation of activated platelets and, in the case of pathological erythrocytes rigidity, deformability and improves their ability to filtration.
Suction. After oral administration Piracetam is rapidly and almost completely absorbed from the gastrointestinal tract. The bioavailability of piracetam is close to 100%. After a single dose of the drug at a dose of 3.2 g of Cmax was 84 mcg / mL after multiple dose 3.2 mg three times a day - 115 .mu.g / ml achieved after 1 h in the blood plasma after 5 hours and - in the cerebrospinal fluid. Ingestion decreases Cmax by 17% and increase the Tmax to 1.5 hours. In women, when receiving a dose of piracetam 2.4 g of Cmax and AUC is 30% higher than in men.
Distribution. It binds to plasma proteins. Vd is about 0.6 liter / kg. Piracetam penetrate the BBB and placental barrier. In animal studies found that piracetam selectively accumulates in the tissues of the brain cortex, predominantly in the frontal, parietal, and occipital lobes in the basal ganglia and cerebellum.
Metabolism. Do not metabolized in the body.
Withdrawal. T1 / 2 4-5 h of blood plasma and 8.5 hours - from cerebrospinal fluid. T1 / 2 is not dependent on the route of administration. 80-100% piracetam excreted by the kidneys in unchanged form by glomerular filtration. Piracetam total clearance in healthy volunteers is 80-90 ml / min. T1 / 2 is prolonged in renal failure (ESRD when - up to 59 hours). Pharmacokinetics piracetam not altered in patients with hepatic failure.
Indications:
- Symptomatic treatment of psycho-organic syndrome, particularly in elderly patients suffering from memory loss, dizziness, decreased concentration and total activity, changes in mood, behavior disorder, gait disorder, as well as in patients with Alzheimer's disease and senile dementia of the Alzheimer type.
- Treatment of the effects of stroke such as speech disorders, disorders of the emotional sphere, to improve motor and mental activity.
- Chronic alcoholism - for the treatment of psycho-organic and withdrawal syndromes.
- The recovery period after the comatose states, including after injuries and cerebral intoxication.
- Treatment of vertigo and associated disorders of balance, with the exception of vertigo of vascular origin and mental.
- In the complex treatment of lower learning ability in children with psycho-organic syndrome.
- For the treatment of cortical myoclonus as mono- or combination therapy.
- In the treatment of sickle-cell anemia.
Contraindications:
- Hypersensitivity to piracetam or pyrrolidone derivatives, and other ingredients;
- Huntington's chorea;
- acute ischemic stroke (hemorrhagic stroke);
- end-stage chronic renal failure.
Suggested Use:
Daily dose - 30-160 mg / kg, the multiplicity of reception - 2-4 times a day. Inside appointed during meals or on an empty stomach; Tablets and capsules should be taken with liquid (water, juice).
- When the symptomatic treatment of psycho-organic syndrome of chronic, depending on the severity of symptoms, administered 1.2-2.4 g, and during the first week - 4.8 g / day.
- In the treatment of the effects of stroke appoint 4.8 g / day.
- During the period of recovery from coma, as well as the perception of difficulties in persons with brain injuries, the initial dose is 9-12 g / day maintenance - 2.4 g / day. Treatment continues for at least 3 weeks.
- When alcohol withdrawal syndrome during the crisis - 12 g / day in 2-3 divided doses. The maintenance dose - 2.4 g / day.
- Treatment of vertigo and associated equilibrium 2,4-4,8 g per day disorders.
- Children learning correction lower dose is 3.3 g / d (about 8 ml oral solution twice a day). Treatment continues throughout the school year.
- When the treatment begins cortical myoclonus with 7.2 g / day every 3-4 days the dose is increased by 4.8 g / d to a maximum dose of 24 g / day. Treatment continues throughout the period of illness. Every 6 months, attempts to reduce the dose or discontinuation of the drug, to prevent the attack of gradually reducing the dose of 1.2 g every 2 days. In the absence of treatment effect or a slight therapeutic effect is stopped.
- When sickle prophylactic daily dose of 160 mg / kg body weight, divided into four equal doses. During the crisis, -300 mg / kg intravenously.
Packaging:
- Comes in original packaging. Item is brand new and unopened.
Storage:
- Keep away from direct sunlight.
- Keep locked and away from children.
- Store in dry place at room temperature.
- Do not exceed storage temperature higher than 25 C
Important notice- the outer box design may vary before prior notice!
Related products


 Cart
Cart