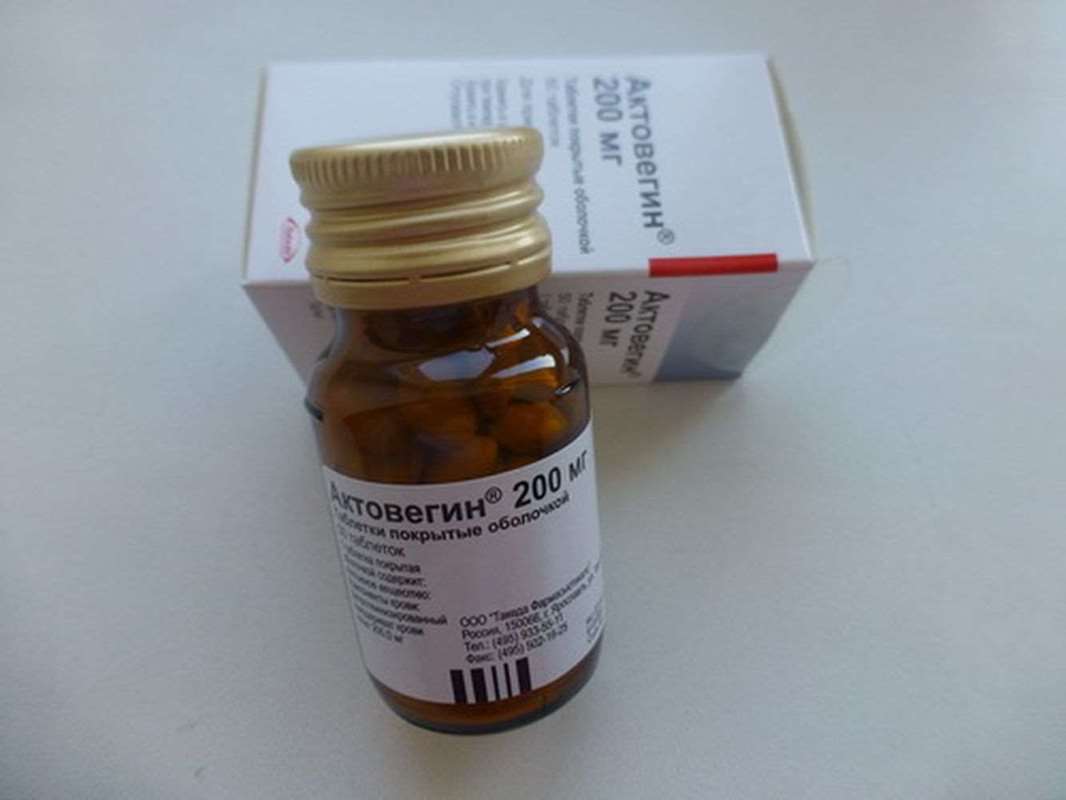
Actovegin - is an extract of calf blood. The drug has not been proven in studies. Some doctors say positive aktovegina application, but it can only be explained by the placebo effect.
Actovegin is highly purified (deproteinized) gemoderivat which is prepared by dialysis and ultrafiltration. The preparation contains the complex micro and macroelements, amino acids, oligopeptides, nucleosides, fatty acids, oligosaccharides - and vital physiological components only. Aktovegin not immunogenic, non-pyrogenic and well it penetrates the blood-brain barrier, as the molecular weight of the constituent components is very low.
Actovegin beneficial effect on the organs and tissues of the body. Aktovegin has a high degree of safety. This allows you to use the drug in patients of different ages, with different concomitant diseases.
Actovegin made in accordance with the safety requirements in relation to prion diseases (infectious agents priony- protein nature, which can be transmitted through the blood).
The main effect aktovegin is its ability to protect organs and tissues from damage in the conditions of insufficient blood supply (anti-ischemic effect). It is associated with the anti-hypoxic and antioxidant mechanisms of drug action.
In addition to the anti-ischemic effect Actovegin has a complex metabolic effects associated with its constituent amino acids, micro and macro elements.
Improving the work of the intracellular enzyme systems, Actovegin strengthens synthetic processes and thus stimulate anabolism. This improves tissue regeneration, repair damage occurs.
Recommendations for use in sport:
- Accelerate the recovery process after stress, aimed at the development of endurance.
- Prevention and treatment of chronic degenerative physical overexertion embodiment of the cardiovascular system (particularly for the purpose of training under the conditions of middle).
- Keeping a high level of performance in a high-altitude climbing.
- Achillodynia and muscle tears.
- Traumatic brain injury.
- The syndrome of cerebral insufficiency boxers.
- Frostbite among representatives of winter sports.
- The increase in body weight.
- Prevention and treatment of sports psevdonefropatii.
Side effect:
Allergic reactions (skin rash, skin redness, hyperthermia), up to anaphylactic shock.
Suggested Use:
B / A / O (including in the form of infusion), in / m.
In connection with the potential development of anaphylactic reactions it is recommended to test for hypersensitivity to the drug prior to infusion.
Ischemic stroke. 250-500 ml for infusion of sodium chloride solution (1000-2000 mg) per day / in over 2 weeks, or 20-50 ml of solution for injection (800-2000 mg) in 0.9 ml 200-300 % sodium chloride solution or 5% dextrose / drip for 1 week, then - 10-20 ml (400-800 mg) / drip daily for 1 week, then - 10-20 ml ( 400-800 mg) / drip for 2 weeks. Then - switch to tablet form.
Metabolic and cerebrovascular disorders. 250-500 ml of solution for infusion (1000-2000 mg) per day and 5-25 ml of solution for injection (200-1000 mg) per day / per day for 2 weeks followed by transfer to or tablet form further - 250 ml / in several times a week (for infusions in dextrose saline).
Peripheral (arterial and venous) vascular disorders and their consequences. 250 mL (1,000 mg) in a solution for infusion / a or a / a day or several times a week, followed by the transition to the tablet form (for infusion of sodium chloride solution). Or 20-30 ml solution for injection (800-1200 mg) in 200 ml of 0.9% sodium chloride or 5% dextrose in / and or / day for 4 weeks.
Diabetic polyneuropathy. 250-500 ml for infusion of sodium chloride solution or 50 ml solution for injection (2000 mg) per day / in for 3 weeks followed by transfer to tablet form - Table 2-3. 3 times a day for at least 4-5 months.
Wound healing. 250 ml solution for infusion (1000 mg) in / daily or several times a week, depending on the rate of healing. Or 10 ml solution for injection (400 mg) in / or in the 5 ml / m daily or 3-4 times a week, depending on the healing process. Perhaps a joint application with Aktovegina� dosage forms for external use.
Prevention and treatment of radiation skin lesions and mucous membranes with radiation therapy. 250 ml / in the day before and daily during radiation therapy, and for 2 weeks after its closure, followed by transfer to tablet form (for infusion of sodium chloride solution) solution for infusion (1000 mg). introduction rate - about 2 ml / min. Or 5 ml of solution for injection (200 mg) / day in breaks in radiation exposure.
For Actovegin solution for injection, in addition
Radiation cystitis. A transurethral, daily, 10 ml of solution for injection (400 mg) in combination with antibiotic therapy. introduction rate - about 2 ml / min.
Duration of treatment is determined individually according to the symptoms and severity of the disease.
Instructions for use ampoules with breaking point
1. Place the tip of the ampule breaking point upwards.
2. Gently tapping his finger and shaking the vial to give a solution from the tip of the ampule to flow down.
3. Break off the tip of the ampoule at fault movement from itself.
Packaging:
- Comes in original packaging. Item is brand new and unopened.
Storage:
- Keep away from direct sunlight.
- Keep locked and away from children.
- Store in dry place at room temperature.
- Do not exceed storage temperature higher than 25 C
Important notice - the outer box design may vary before prior notice!

 Cart
Cart