Doctor Doping Shop
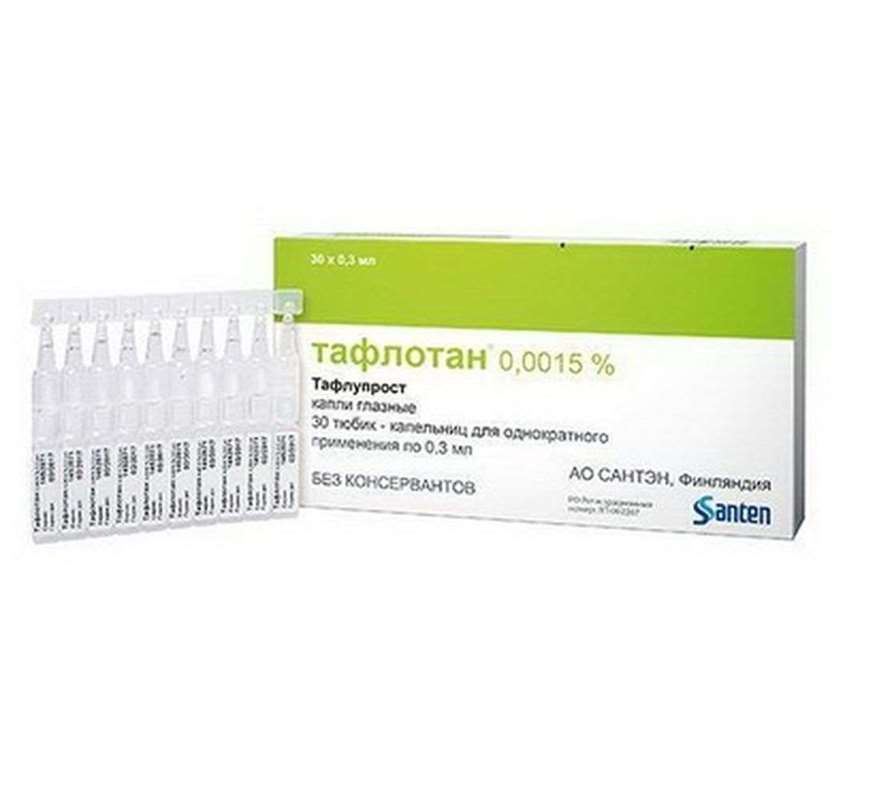
Taflotan eye drops 0.0015% 0.3ml 30 pieces
USD 74.99
In stock
Be the first to review this item
Taflotan eye drops - a drug used in ophthalmology for the treatment of glaucoma. The main active substance of the drug is tafluprost, which has a persistent therapeutic effect, normalizing intraocular pressure. Suitable for permanent use.
Tafluprost is a fluorinated analogue of prostaglandin F2α. The acid of tafluprost, being its biologically active metabolite, has a high activity and selectivity for the human FP-prostanoid receptor. The affinity of tafluprost acid for the FP receptor is 12 times higher than the affinity of latanoprost. Pharmacodynamic studies in monkeys have shown that tafluprost reduces intraocular pressure, enhancing the outgrowth outflow of aqueous humor.
Pharmacodynamic effect
Experiments on monkeys with normal and increased intraocular pressure (IOP) demonstrated that Tafluprost is an effective drug for reducing IOP. In a study on the IHD-lowering effect of tafluprost metabolites, it was found that only tafluprost acid significantly reduces IOP.
Studies on rabbits treated for 4 weeks with an ophthalmic tufluprost solution, 0.0015%, once a day, showed that blood flow in the optic nerve disk significantly (by 15%) increased in comparison with baseline, when measured at 14 and 28 day using laser speckle of flowery.
Clinical effect
Decrease in intraocular pressure begins within 2-4 hours after the first installation of the drug, and the maximum effect is achieved after about 12 hours. The duration of the effect is maintained for at least 24 hours. Leading studies on the use of tafluprost containing a benzalkonium chloride preservative have shown that tafluprost is effective as a monotherapy, and also has an additive effect when applied as an adjunct therapy to timolol. In a 6-month study, tafluprost, at various time points during the day, demonstrated a significant, IOP-lowering effect: 6 to 8 mm Hg. in comparison with latanoprost, which reduces IOP by 7-9 mm Hg. In another 6-month clinical trial, tafluprost reduced IOP by 5-7 mm Hg, and timolol 4-6 mm Hg. The IOP-lowering effect of tafluprost was also maintained with an increase in the duration of these studies to 12 months. In a 6-week study, the IOP-lowering effect of tafluprost was compared to the effect of its indifferent excipient when used in conjunction with timolol. Compared with the baseline (measurement was performed after a 4-week course of treatment with timolol), the additional IHD-lowering effect was 5-6 mm Hg. in timolol-tafluprost group and 3-4 mm Hg. in the group timolol-iidiferentny filler. In a small cross-sectional study, with a 4-week treatment period, dosage forms with a preservative and no preservative showed a similar IHD-lowering effect - more than 5 mmHg.
In addition, in a 3-month study in the United States when compared, the composition of tafluprost without a preservative with timolol. as well as without a preservative, it was found that tafluprost reduced IOP by 6.2-7.4 mm Hg. at different time points, whereas the values for timolol varied between 5.3 and 7.5 mm Hg.
Pharmacokinetics
Absorption
After the installation of eye drops tafluprost, 0.0015% in a dropper tube, without preservative, once a day, one drop in both eyes for 8 days, its plasma concentrations were low and had a similar profile on days 1 and 8. The plasma concentrations reached a maximum 10 minutes after the installation, and decreased to a level lower than the lower detection limit (10 pg / ml) less than an hour after the administration of the drug. The mean Cmax values (26.2 and 26.6 pg / ml) and AUCo-last (394.3 and 431.9 pg / min / ml) were almost the same on days 1 and 8, indicating that during the first week of treatment, a stable concentration of the drug. Between drug forms with a preservative and without a preservative, no statistically significant differences in systemic bioavailability were found.
In a rabbit study, the absorption of tafluprost into watery moisture, after a single installation of an ophthalmic solution of tafluprost, 0.0015% with a preservative and no preservative, was comparable.
Distribution
In a monkey study, no specific distribution of radiolabeled tafluprost was observed in the iris, ciliary body, or in the choroid of the eye, including the retinal pigment epithelium, indicating a low affinity for the drug to the melanin pigment.
Autoradiography in rats showed that the highest concentration of radioactivity was observed in the cornea, followed by eyelids, sclera, and the iris. Systemic radioactivity spread to the tear apparatus, the sky, esophagus, gastrointestinal tract, kidneys, liver, gallbladder and urinary bladder. The binding of tafluprost acid to human serum albumin in vitro is 99% for 500 ng / ml of tafluprost acid.
Biotransformation
The main way of metabolism of tafluprost in the human body, tested in vitro is hydrolysis with the formation of a pharmacologically active metabolite, an acid of tafluprost, which is then masturbated
Indications:
- to reduce increased intraocular pressure in patients with open-angle glaucoma and ophthalmic hypertension;
- as monotherapy in patients who are shown eye drops that do not contain a preservative, or with insufficient response to first-line drugs, or who do not tolerate first-line drugs or who have contraindications to these drugs;
- as an additional therapy for beta-blockers.
Contraindications:
Hypersensitivity to tafluprost or any of the inert fillers of Taflotan.
Special instructions:
Before the treatment, patients should be warned about the possibility of excessive eyelash growth, darkening of the eyelid skin and increased pigmentation of irises. Some of these changes may be permanent, and this can lead to differences in the appearance of the eyes, if only one eye is treated.
The change in the pigmentation of the iris occurs slowly and may not be noticeable for several months. The color changes of the eyes are observed mainly in patients with irises of mixed colors, for example, if the eyes are brownish-blue, gray-brown, yellow-brown or green-brown. Risky lifetime heterochromia is suspected if only one eye is treated.
There is no experience with tafluprost in cases of neovascular, occlusive, narrow-angle or congenital glaucoma. There is only limited experience with tafluprost in patients with aphakia, pigmentary or pseudoexfoliation glaucoma.
It is recommended to be careful when treating patients with aphakia, artifacts, damaged posterior lens capsule or anterior chamber lens or patients with established risk factors for cystoid edema of maculolytic iritis / uveitis.
There is no experience of using the drug in patients with heavy asthma. In this regard, patients of this group should be treated with consistency.
It was reported that benzalkonium chloride, which is usually used as a preservative in ophthalmic preparations, causes spotkeratopathy and / or toxic ulcer keratopathy. Since tuflotan vflacconi contains benzalkonium chloride, careful monitoring is necessary in cases of frequent or prolonged use in patients with dry eyes, and also in cases where the cornea is at risk.
Benzalkonium chloride, can also cause eye irritation and the discoloration of soft contact lenses. Avoid contact with soft contact lenses. Remove contact lenses before using the drug and insert them again not earlier than 15 minutes after instillation.
Impact on the ability to drive vehicles and manage mechanisms
Tafluprost does not affect the ability to drive a car and work with machinery. As with any other ophthalmic device, a short-term blurring of vision may occur after the preparation is installed. In this case, the patient should wait until vision is restored, and only then drive or operate mechanical equipment.
Suggested Use:
The recommended dose is one drop of eye drops of Taflotan in the conjunctival sac of the affected eye (eye) once a day, in the evening.
The dose should be installed strictly once a day, as more frequent use can reduce the effect of reducing intraocular pressure.
Only for single use. The contents of one tube-dropper is sufficient for burying in both eyes. The remaining drug should be discarded immediately after use.
Application in elderly patients
In the treatment of elderly patients, there is no need for dose changes.
Application in children and adolescents
Safety and effectiveness of Tafluprost in children and adolescents under the age of 18 years is not established. No data available.
Use in cases of impaired kidney / liver function
Studies on the effects of tafluprost on patients with renal / hepatic disorders have not been carried out, so care should be taken when treating this category of patients.
Method of application
To reduce the risk of darkening the skin of the eyelids, patients should remove excess solution from the skin. As with the application of other eye drops, nasolacrimal occlusion is recommended - a soft eyelid closure after the drug has been installed. This can reduce the systemic absorption of medications injected through the eyes.
When using several topical ophthalmic drugs, the intervals between their use should be at least 5 minutes.
Packaging:
- Comes in original packaging. Item is brand new and unopened.
Storage:
- Keep away from direct sunlight.
- Keep locked and away from children.
- Store in dry place at room temperature.
- Do not exceed storage temperature higher than 25 C
Important notice- the outer box design may vary before prior notice!
Related products


 Cart
Cart