Doctor Doping Shop
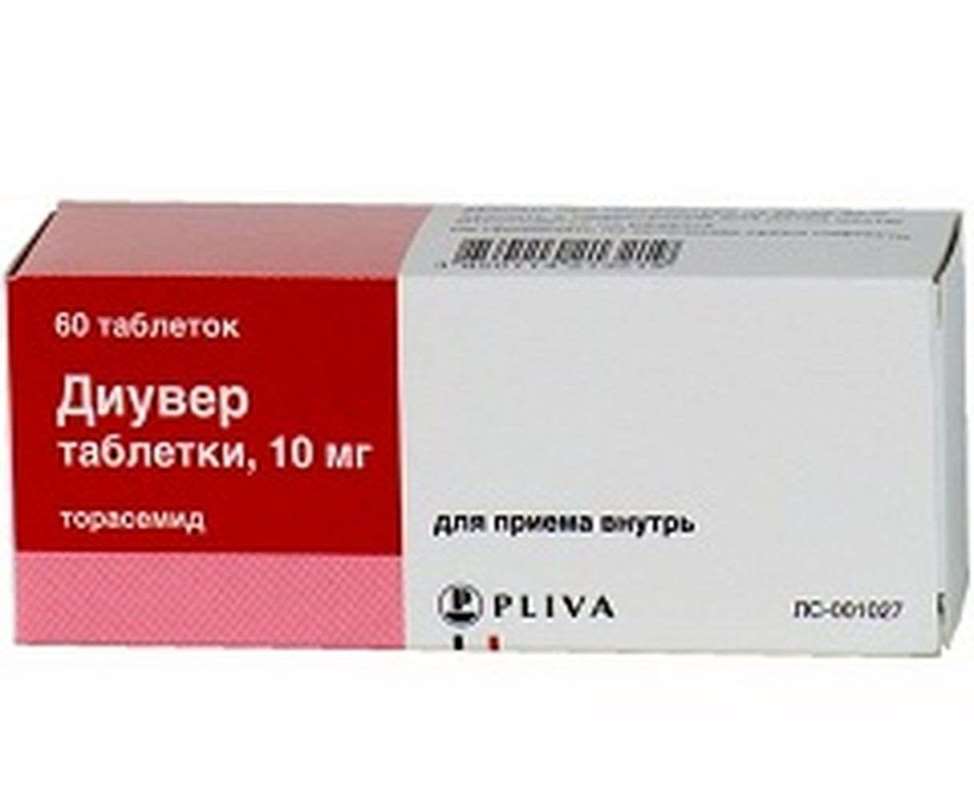
Diuver 10mg 60 pills
USD 93.99
In stock
Be the first to review this item
Diuver (Torasemidum, Torasemide) - is a loop diuretic. The maximum diuretic effect develops after 2-3 hours after ingestion of the drug. The main mechanism of action of the drug due to the reversible binding of torasemide kontransporterom sodium / chlorine / potassium ions located in the apical membrane of the thick ascending part of Henle segment loop, resulting in reduced or completely inhibited by sodium ion reabsorption and decreases the osmotic pressure of the intracellular fluid and water reabsorption. Blocks myocardial aldosterone receptors, reduces fibrosis and improves diastolic myocardial function.
Torasemide less than furosemide causes hypokalemia, while it is more active, and more lasting effect.
The use of torasemide is the most reasonable choice for long-term therapy.
After oral torasemide is rapidly and almost completely absorbed in the digestive tract. Cmaxtorasemida plasma observed after 1-2 hours after ingestion after eating. Bioavailability is 80-90% with slight individual variations.
The diuretic effect persists for up to 18 hours to facilitate the portability of therapy due to lack of very frequent urination during the first hours after ingestion of the drug, which limits patient activity.
Communication to plasma proteins up to 99%. Visible Vd is 16 liters.
It is metabolized in the liver via the cytochrome P450 isoenzyme system. As a result, sequential reactions of oxidation and hydroxylation are formed three annular hydroxylation metabolite (M1, M3 and M5) that bind to plasma proteins at 86, 95 and 97%, respectively.
T1 / 2 of torasemide and its metabolites is 3-4 hours, and does not change with CRF. Torasemide total clearance of 40 ml / min, renal clearance - 10 ml / min. On average, about 83% of the dose excreted by the kidneys: in unchanged form (24%) and mainly in the form of active metabolites (the M1 - 12%, M3 - 3%, M5 - 41%).
In renal insufficiency, T1 / 2 is not changed, T1 / 2 and M5 metabolites MH increases. Torasemide and its metabolites are excreted in slightly by hemodialysis and hemofiltration.
In liver failure plasma concentration increases due to the reduction of torasemide drug metabolism in the liver. In patients with cardiac or hepatic insufficiency T1 / 2 of torasemide and metabolite M5 significantly increased accumulation of the drug is unlikely.
Testimony:
Edematous syndrome of various genesis, including in chronic heart failure, diseases of the liver, lungs and kidneys; arterial hypertension.
Contraindications:
- Hypersensitivity to torasemide or any of the components;
- allergic to sulfonamides (sulfa antimicrobials or sulfonylureas);
- renal failure with anuria;
- hepatic coma and precoma;
- severe hypokalemia;
- severe hyponatremia;
- hypovolemia (hypotension with or without), or dehydration;
- pronounced violations of the outflow of urine from any cause (including unilateral urinary tract disease);
- glycoside intoxication;
- acute glomerulonephritis;
- decompensated aortic and mitral stenosis, hypertrophic obstructive cardiomyopathy;
- elevated central venous pressure (more than 10 mm Hg..);
- hyperuricemia;
- lactose intolerance, lactase deficiency or glucose-galactose malabsorption;
- lactation;
- age 18 years.
With caution: arterial hypotension; constrictive atherosclerosis of cerebral arteries; hypoproteinemia; predisposed to hyperuricemia; violation of the outflow of urine (benign prostatic hyperplasia, narrowing of the urethra or hydronephrosis); ventricular arrhythmia in history; acute myocardial infarction (increased risk of cardiogenic shock); diarrhea; pancreatitis; diabetes mellitus (impaired glucose tolerance); hepatorenal syndrome; gout; anemia; pregnancy.
Special instructions:
should carry out the correction of water and electrolyte balance Diuver Prior to his appointment. Precautions taken for gout or inclination to increase uric acid levels. Prolonged treatment Diuver recommended to control electrolyte balance, glucose, uric acid, creatinine and lipids in the blood.
In patients with diabetes mellitus type 1 or 2 must regularly monitor the level of glucose in the blood.
During treatment, patients should avoid driving and other activities that require high concentration and speed of psychomotor reactions.
Suggested Use:
Once a day, after breakfast, washed down with a little water.
Edematous syndrome of various genesis, including in CHF, liver, lungs and kidneys
The usual therapeutic dose is 5 mg orally once daily. If necessary, the dose should be gradually increased up to 20-40 mg once a day, in some cases - to 200 mg per day. The drug is prescribed for a long period or until disappearance of edema.
Arterial hypertension
The starting dose is 2.5 mg (2.1 to 5 mg tablets) once daily. If necessary, the dose may be increased up to 5 mg once a day.
Elderly patients Dose adjustment is not required.
Side effects:
From the water-electrolyte and acid-base balance: hyponatremia, hyposalemia, hypokalemia, hypomagnesemia, hypocalcemia, metabolic alkalosis. Symptoms indicating the development of disorders of electrolyte and acid-base balance state can be a headache, confusion, cramps, tetany, muscle weakness, heart rhythm disturbances and dyspepsia; hypovolemia and dehydration (often in elderly patients), which may cause hemoconcentration with a tendency to develop thrombosis.
From the CCC: an excessive fall in blood pressure, orthostatic hypotension, collapse, tachycardia, arrhythmias, decreased CBV.
From a metabolism: hypercholesterolemia, hypertriglyceridemia; transient increase in the concentration of urea and creatinine in the blood; increased concentration of uric acid in the blood, which may cause or exacerbate symptoms of gout; impaired glucose tolerance (can be a manifestation of latent diabetes mellitus flowing).
From the urinary system: oliguria, acute urinary retention (such as prostatic hyperplasia, narrowing of the urethra, hydronephrosis); interstitial nephritis, hematuria, reduced potency.
On the part of the digestive tract: nausea, vomiting, diarrhea, intrahepatic cholestasis, elevated liver enzymes, acute pancreatitis.
On the part of the central nervous system, organ of hearing: hearing loss, usually reversible, and / or ringing in the ears, especially in patients with renal failure or hypoproteinemia (nephrotic syndrome), paresthesia.
For the skin: itching, urticaria, other rashes or bullous skin lesions, erythema multiforme, exfoliative dermatitis, purpura, fever, vasculitis, eosinophilia, photosensitivity; severe anaphylactic or anaphylactoid reactions up to shock, which have hitherto been described only after the on / in the introduction.
From the peripheral blood: thrombocytopenia, leukopenia, agranulocytosis, aplastic or hemolytic anemia.
Drug interactions:
In a joint application torasemide increase myocardial sensitivity to cardiac glycosides (because of the possible development of hypokalemia and hypomagnesemia).
With simultaneous use of torasemide with mineral and glucocorticoids and laxatives may increase potassium excretion.
With simultaneous use of torasemide enhances the effect of antihypertensive drugs.
In a joint application Torasemide, especially at high doses, may potentiate nephrotoxicity and ototoxic effects of aminoglycoside antibiotics group, cisplatin toxicity, nephrotoxic effects of cephalosporins and cardio- and neurotoxic effects of lithium products.
In a joint application torasemide may increase the effects of peripheral muscle relaxants (coumarin derivatives) and theophylline.
With simultaneous use of torasemide with salicylates (in high doses) their toxic effects may be intensified.
In a joint application Torasemide may reduce the efficacy of hypoglycemic (anti-diabetic) agents.
The sequential or simultaneous administration of torasemide with ACE inhibitors may transient drop in blood pressure. If necessary, use a combination of whether or reduce the initial dose of ACE inhibitor, or to reduce the dose of torasemide (or temporarily canceling it).
In a joint application of NSAIDs and probenecid may reduce the diuretic and hypotensive effect of torasemide.
Cholestyramine can reduce absorption from the digestive tract of torasemide (according to the pre-clinical study in animals).
Packaging:
- Comes in original packaging. Item is brand new and unopened.
Storage:
- Keep away from direct sunlight.
- Keep locked and away from children.
- Store in dry place at room temperature.
- Do not exceed storage temperature higher than 25 C
Important notice- the outer box design may vary before prior notice!
Related products


 Cart
Cart